Hantan | Hondius
On the state of virology, pandemic preparedness, and the incentives research runs on.
Research & Culture | Science & Art | Literature
IV. This isn’t COVID - the biology
I. Hantan, 1951.
In the spring of 1951, a young soldier in the central Korean front collapsed with a fever no one could explain.
He had been stationed near a tributary of the Han River — a narrow, fast-moving river the locals called the Hantan. The river ran through what is now the Iron Triangle, the most fiercely contested ground of the war, and the soldier had spent his nights in a bunker burrowed into a hillside that crawled with rats. They were everywhere, in the trenches and the supply caches and the food stores. No one paid them much mind. There was a war on.
Within days the soldier was bleeding from his gums. His blood pressure cratered. His kidneys stopped. He died in a field hospital with no name for what had killed him.
By the end of that summer, more than 800 UN soldiers in his sector had been hospitalized with the same disease. By the end of the war, the count had reached 3,200. About one in ten died.1 The US Army, faced with a mystery that wasn’t malaria, wasn’t typhus, wasn’t anything in the field manual, set up a dedicated hemorrhagic fever center near the front. They called the disease Korean hemorrhagic fever. They could describe it down to the petechiae on the soldiers’ chests. They could not isolate it. The cause was, in their words, a rickettsial agent of unknown origin — a phrase that meant nobody had any idea.
Some doctors thought it was a toxin. Some thought it was a fungus. Some thought it was the rats.
The doctors who thought it was the rats were right. It would take twenty-five years for anyone to prove it.
In 1976, a South Korean virologist named Ho Wang Lee — working in cramped lab conditions at Korea University, on a small research budget — finally isolated an antigen from the lungs of striped field mice trapped along the Hantan River.2 The mice were infected. They had always been infected. The soldiers had been breathing in their excrement for two and a half years. The virus was named after the river: Hantaan virus. The genus that grew up around it — every hantavirus discovered since, on every continent — bears the name of a Korean tributary, where someone finally bothered to look.
Hold that picture.
This week, a Dutch cruise ship called the MV Hondius is sailing across the Atlantic with three dead passengers, eight infected, a critically ill ship’s doctor evacuated to a European hospital, a KLM flight attendant in an isolation ward in Amsterdam, and around 146 people still on board — refused entry by Cape Verde, refused dock by the Canary Islands’ regional president, headed for Tenerife under the protection of a Spanish national government that has overruled him.
The disease they are carrying is a descendant of the same family of viruses that killed the soldiers along the Hantan, separated by seventy-five years and a continent. It is not new. It is not surprising. It is not, with the data we have now, a pandemic.
It is, however, a near-miss. And the system catching it has not learned anything from the last one.
What follows is the attempt to make sense of it all. Where the facts are, where the science is, where the gaps are — and what the entire situation says about the systems we use to fund, conduct, and operate pandemic preparedness.
II. Vocabulary
The words being used interchangeably this week are not interchangeable. I generally find it helpful to establish this baseline.
An outbreak is a sudden cluster of cases of a disease in a specific place, beyond what is normally expected. Eight cases on a cruise ship is an outbreak.
An epidemic is a disease spreading actively through a population in a region or country, beyond baseline. Argentina’s 101 hantavirus cases since June 2025, roughly double the prior year’s caseload, is approaching epidemic territory for that country.
A pandemic is an epidemic that has spread across multiple countries or continents with sustained human-to-human transmission. The threshold is in the last clause. COVID-19 met it. The 1918 flu met it. HIV/AIDS met it. The Hondius does not.
Endemic is the baseline state. The disease is constantly present at predictable levels in a population. Malaria is endemic across much of sub-Saharan Africa. Hantavirus is endemic in deer mice across the Americas and in striped field mice across East Asia. It has been endemic in those rodent populations longer than humans have lived in those places.
The word “pandemic,” when it shows up in a headline this week, is being used loosely. It has a technical meaning, and the Hondius does not meet it. What the world is watching is an outbreak with secondary transmission risk under investigation as a possible multi-country epidemic.
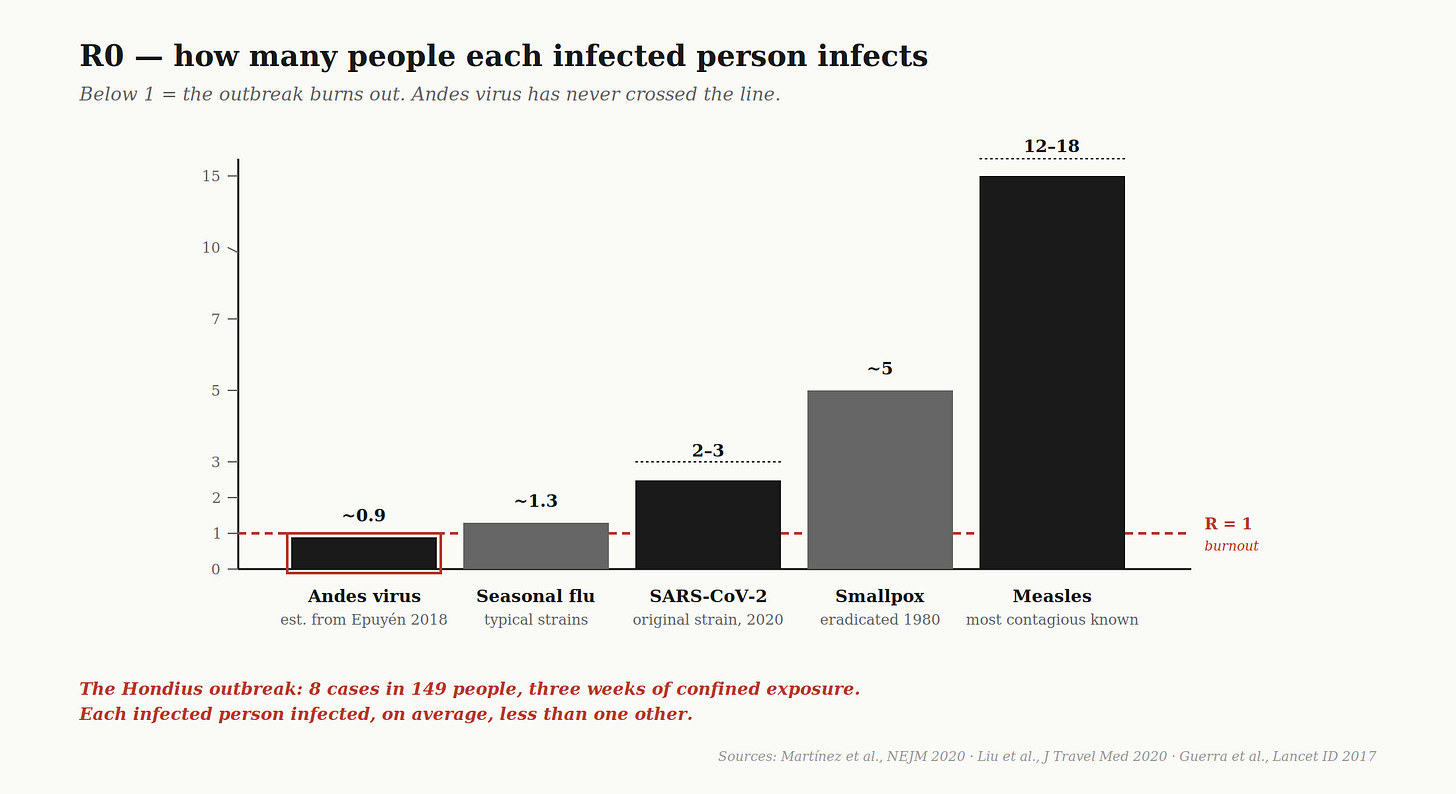
The second piece of vocabulary — and the more important one — is R: the basic reproduction number. It indicates the average number of people one infected person will go on to infect, in a fully susceptible population, under specified conditions. If R0 is greater than 1, the outbreak grows. If R0 is less than 1, it burns out. If R0 equals 1, it stays put.
There are two R0s; they are not the same number. Conflating them is how a reader ends up thinking a virus is more dangerous than it is, or — worse — less.
Call the first one intrinsic R0. This is what the virus is capable of, biologically. It comes from controlled studies, animal models, and clean outbreak data, and it answers the question: if everything else is held constant, how does this pathogen want to spread? For Andes virus, the intrinsic R0 estimated from the worst documented super-spreader event — Epuyén, Argentina, 2018, which we will get to — sits below 1.0.3 For original-strain SARS-CoV-2, it was 2 to 3. For measles, it is 12 to 18. This is the number the biology hands you.
Call the second one competency R04 This is what actually happens once human behavior, policy decisions, communication failures, and institutional dysfunction get layered on top. Competency R0 can be higher or lower than intrinsic R0 depending on whether the people in charge are doing their jobs.
COVID’s intrinsic R0 was 2 to 3. Its competency R0, in the worst-managed regions in 2020, spiked to 4 to 6 — because messaging was contradictory, because containment policy flip-flopped, because asymptomatic transmission was not taken seriously, because testing was rationed, because federalism meant there were fifty different protocols across states.
This distinction matters because the data from the Hondius tells us the intrinsic R0 of Andes virus is doing exactly what it should (consistent with 30 years of data): burning out below 1. The competency R0 is being inflated by every governance failure stacked on top of it. For instance, there was a twenty-one-day gap between the first death and the first WHO notification. Cape Verde is refusing dock. Tenerife is refusing dock. Thirty passengers disembarked before containment was in place. Those aren’t viral decisions.
III. The Hondius
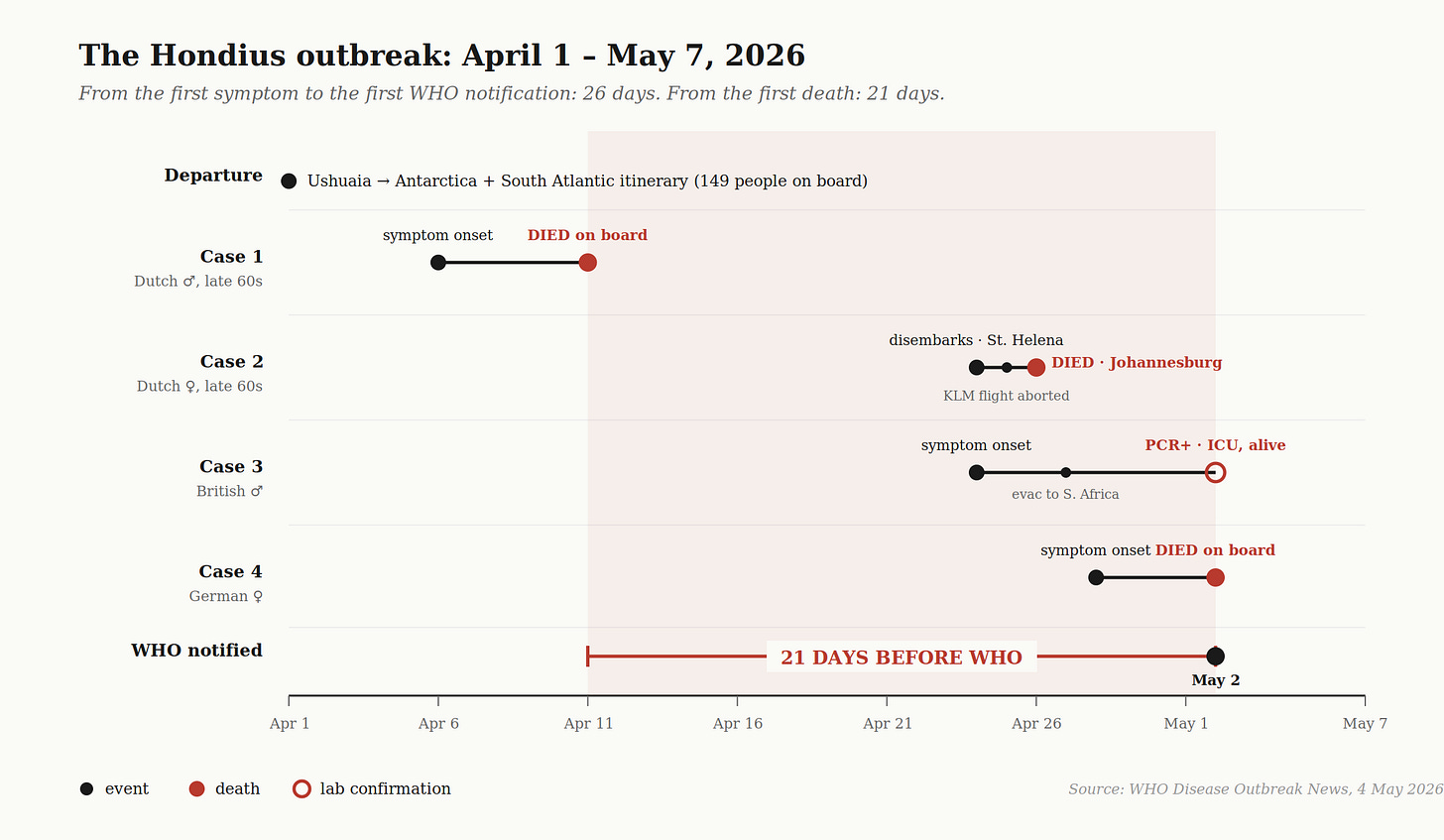
The MV Hondius is a Dutch-flagged polar expedition vessel built in 2019 by Brodosplit in Croatia, operated by Oceanwide Expeditions. It carries up to 196 passengers and 72 crew. On April 1, 2026, it left Ushuaia, Argentina — the southernmost city in the world — with 147 people on board representing 23 nationalities. Spanish, British, and American passengers; mostly Filipino crew. Its route was to Antarctica and the remote South Atlantic — South Georgia, Tristan da Cunha, Saint Helena, Ascension. None of the passengers showed symptoms at departure.
Argentine investigators believe the first two cases — a Dutch couple in their late sixties — contracted Andes virus during a four-month road trip through Chile, Uruguay, and Argentina from late November 2025 through the day they boarded the ship. The leading hypothesis, per anonymous Argentine investigators who spoke to AP, is that exposure happened during a birdwatching outing near Ushuaia, possibly at a landfill where rodents would have been present. The Argentine health ministry is now trapping rodents along the couple’s route and running 2,500 diagnostic tests.
What happened next:
April 6. Case 1, the Dutch husband, develops fever, headache, abdominal pain, diarrhea. The ship is somewhere in the Antarctic Peninsula.
April 11. Case 1 dies on board. He has been dead for thirteen days before any laboratory testing happens. No microbiological samples are taken. The body is held on the ship.
April 24. The ship reaches Saint Helena. Case 1’s body is removed. His wife — Case 2 — disembarks with gastrointestinal symptoms. Thirty other passengers also disembark at Saint Helena. They scatter to at least 12 countries: 7 British, 6 American, plus Canadian, German, Singaporean, Turkish, Swiss, and other nationalities.
April 24. Case 3, a British male, presents to the ship’s doctor with fever, shortness of breath, and signs of pneumonia.
April 25. Case 2 boards a KLM flight from Johannesburg to Amsterdam. The cabin crew sees how sick she is, escorts her off the aircraft before takeoff, and the flight departs without her. Her brief presence on the plane is long enough for one flight attendant to attend to her.
April 26. Case 2 dies on arrival at the Johannesburg emergency department.
April 27. Case 3 is medically evacuated from Ascension Island to South Africa.
April 28. Case 4, a German woman, develops symptoms.
May 2. Case 4 dies on board. South Africa’s National Institute for Communicable Diseases confirms hantavirus in Case 3 by PCR. The WHO is notified for the first time. It has been twenty-one days since Case 1 died and twenty-six days since the first symptom onset.
May 3. The ship docks in Praia, Cape Verde.
May 4. PCR confirms hantavirus in Case 2.
May 5–6. The strain is identified as Andes virus. A KLM flight attendant in Amsterdam is hospitalized with mild symptoms after her brief contact with Case 2. A Swiss case is identified — a passenger who had disembarked earlier and gone home. Three people are evacuated from the ship in Cape Verde, including the ship’s doctor.
May 6. Cape Verde has done what it can. The ship leaves for Tenerife. The Canary Islands’ regional president, Fernando Clavijo, refuses entry, citing COVID-era trauma. Spain’s national government overrules him. The WHO calls it a moral and legal obligation.
May 7 (date of writing). Five confirmed cases, three suspected, three deaths — one definitively confirmed as hantavirus by PCR; the other two presumed but still under investigation. Around 146 people on board. Repatriation is expected to begin May 11 in Tenerife.
There are three things about these numbers to flag.
The attack rate aboard the ship is roughly five percent. Eight cases out of 147 people, in a confined space, sharing food and ventilation for three weeks. If this had been COVID-19 in 2020, the expected attack rate by day 21 would have been 50 to 90 percent. The Diamond Princess — the famous early COVID cruise ship outbreak — ended at 19 percent infected, and that was with a quarantine in place. The Hondius, with no quarantine until very late, is producing a quarter of that rate.
However, the most informative data has not been collected yet. The median incubation period for Andes virus is 18 days, with a documented range of 4 days to 8 weeks. Thirty passengers disembarked at Saint Helena on April 24. Today is May 7. That is thirteen days since disembarkation. The median symptom-onset window for any infected person from that group is May 12, not today. Anyone telling you the outbreak is over is reading data we do not yet have. The window that matters — the window where the secondary cases, if there are going to be secondary cases, will appear — is May 12 through May 20. Until it closes, the honest answer is we are watching.5
The KLM flight attendant is the only documented secondary transmission outside the ship. Her exposure was a brief in-cabin interaction with a critically ill patient before that patient was deplaned. This is consistent with everything known about Andes virus — close, prolonged contact, often during the prodromal phase. It is not consistent with a respiratory virus jumping casually between strangers.
There are eight cases, three dead, one in ICU in Johannesburg, one in Switzerland, one hospitalized in Amsterdam, three aboard the ship. One strain identified, an attack rate consistent with intrinsic R0 below 1, an incubation window that has not closed.
There is also a containment system that took twenty-one days to issue its first alert, watched two governments refuse dock, and is now scrambling to trace passengers across twelve countries.
IV. This isn’t COVID — the biology
The question being asked most often this week, in some form, is: how can anyone be sure this won’t become a pandemic? What if it mutates? What if it goes airborne? Weren’t they saying the same things about COVID in early 2020?
The honest answer is that no virus is at zero pandemic potential. RNA viruses mutate. Hantaviruses, in particular, carry segmented genomes — three separate RNA segments encoding different proteins, called L (the polymerase, the copying machine), M (the surface glycoproteins, which decide what cells the virus can enter), and S (the nucleocapsid). Segmented genomes can do something single-segment viruses cannot: they can reassort. If a single host is co-infected with two different hantavirus lineages, the segments can mix and match into chimeric viruses that have never existed before. This is how flu produces pandemic strains. The 2009 H1N1 swine flu was a reassortant of swine, avian, and human flu segments.
So in principle, yes, hantavirus has evolutionary tools available. In principle.
The more useful answer is structural. SARS-CoV-2 and Andes virus use different doors into the body, attack different tissues, and produce different diseases. They determine everything that matters about transmission and lethality.
SARS-CoV-2 binds to ACE2 receptors. ACE2 is densely expressed in the nose, the throat, and the upper airway. The virus replicates in epithelial cells lining the respiratory tract — the same cells in direct contact with the air being breathed. Every breath, cough, and sneeze aerosolizes virus. The biology and the transmission route are designed for each other. This is why COVID spread on subways and in grocery stores.
Andes virus binds to integrins6 — specifically αvβ3 and αvβ5 integrins, which are adhesion molecules expressed primarily in vascular endothelium, the inner lining of blood vessels. Andes virus does not replicate in airway epithelium. It replicates in the cells lining the blood vessels.
This difference matters quite a bit.
Where the disease happens. COVID kills airway cells, causing inflammation, fluid accumulation in the alveoli, and gas exchange failure. The patient dies of low oxygen. Andes virus damages the vascular endothelium, which makes blood vessels leaky. Fluid floods the lungs not because the virus is there, but because the vessels there are damaged. Blood pressure crashes. The patient dies of shock, not hypoxia.
How fast it kills. COVID’s progression is a 7-to-14-day arc with gradual respiratory decline. There is time to seek care, time to be intubated, time to recover. Andes virus produces a 3-to-5-day prodrome that feels like flu, then a catastrophic 24-to-48-hour cardiopulmonary collapse. Case 1 on the Hondius died five days after his first symptoms.
Why it doesn’t spread well. Because the virus replicates in deep vascular tissue and not in the airway, viral shedding into respiratory secretions is mechanistically lower. The virus is not positioned to exploit the respiratory route. Combined with the fact that infectiousness concentrates in the prodromal phase — when patients look like they have the flu but are not yet visibly critical — and that severe symptoms develop fast enough to put people in hospital beds rather than on subways, the result is the pattern observed for thirty years: chains of two or three transmissions, mostly within families or healthcare settings, that burn out within weeks.
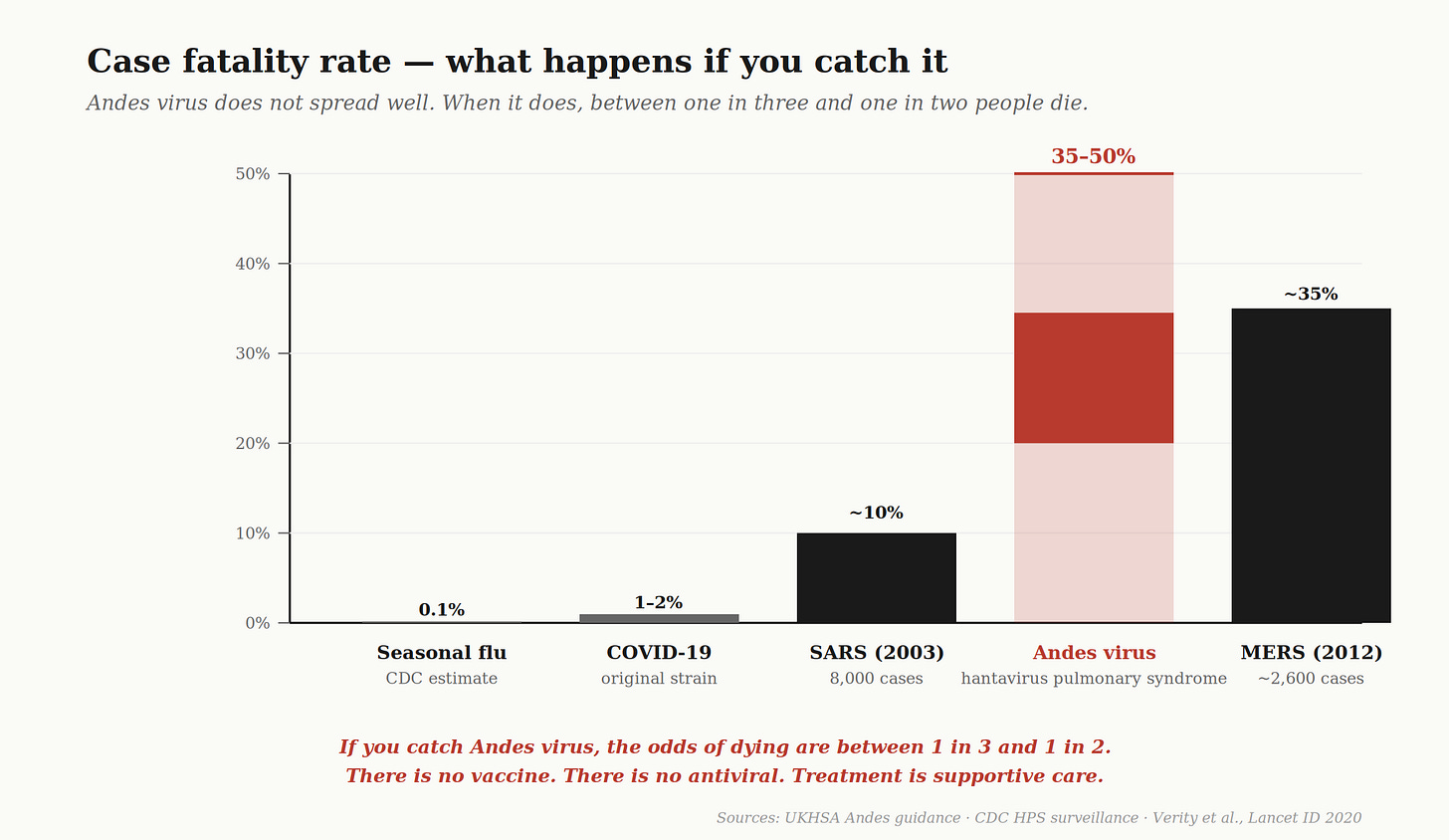
Why mortality is so high. Vascular collapse is harder to treat than respiratory failure. A patient with damaged lungs can be mechanically oxygenated. A patient with a leaking vascular system cannot be easily repaired. Treatment is supportive — vasopressors to raise blood pressure, careful fluid management, and in the worst cases, ECMO, a temporary artificial heart-and-lung machine. The British male in Johannesburg is alive because he reached a hospital with ECMO capability. The Dutch couple died because they were in transit when they hit the cardiopulmonary phase.
For Andes virus to become pandemic-capable, it would need to acquire a fundamentally different receptor binding profile — abandoning vascular integrins for upper-respiratory receptors — shift its replication site from endothelium to airway epithelium, and remain stable in respiratory secretions long enough to transmit casually. This is not a single point mutation. These require complex changes to surface glycoproteins and tissue tropism. The virus has had three decades and multiple super-spreader events to attempt them. It did not.
People asking about COVID are right to be skeptical when they hear this isn’t COVID — they heard this isn’t COVID four months before COVID was, well, COVID. The difference between January 2020 and May 2026 is primarily related to the data that we have.
In January 2020, the world had a novel coronavirus. There were forty-one cases in Wuhan. No confirmed human-to-human transmission yet. A pathogen family that had previously caused two regional epidemics — SARS in 2003 and MERS in 2012 — but had never produced a true pandemic. The WHO’s initial assessment that COVID was probably containable was based on the prior coronavirus track record. It was wrong, but given the data at the time, not unreasonable. What was unreasonable was the institutional inertia in updating it. By mid-January, there was clear human-to-human transmission. By late January, cases on multiple continents. The failure was not the initial assessment. The failure was the lag in revising it.
Andes virus is not novel. We have three decades of full-genome sequences. Hundreds of documented cases. Characterized super-spreader events. Animal models. Receptor-level understanding of how it gets into cells, where it replicates, how it sheds, how it transmits. We know what this virus is. All of it hangs in how we respond to it now.
My cleanest framing of the COVID comparison is this:
You are more likely to catch COVID. Less likely to die from it. You are less likely to catch hantavirus. Far more likely to die from it.
COVID’s lethality came from scale. It infected billions and killed roughly 1-2 percent of the people it infected. Hantavirus’s lethality is intrinsic — small numbers, brutal per-case mortality 7 The threat from this outbreak is not whether it has the potential to become a pandemic, because that will be decided largely by how we respond and how swiftly we act.
V. The river, again
The soldier on the Hantan died in 1951.
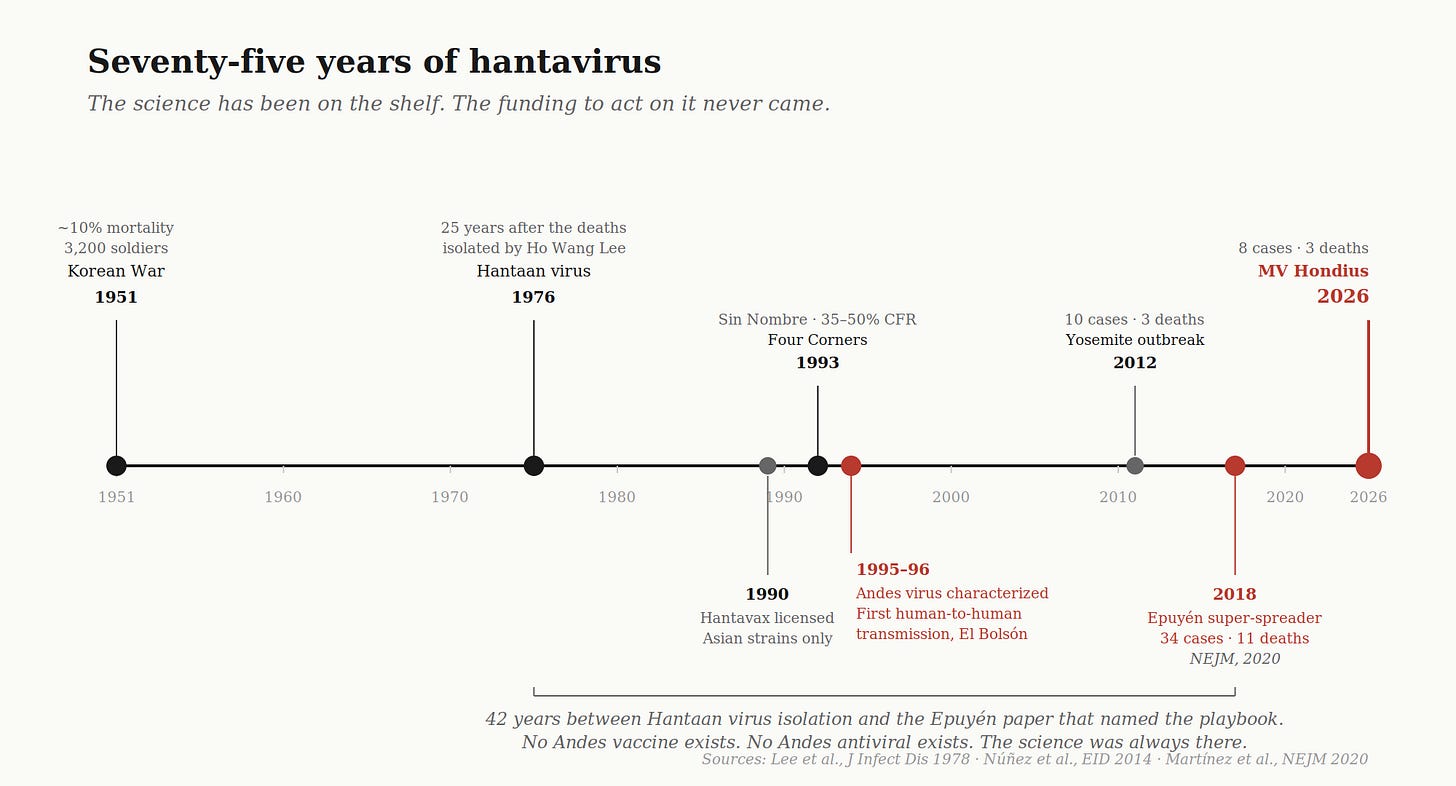
The virus that killed him was named in 1976, twenty-five years later, because a single Korean researcher refused to stop looking, plus the rats nobody else had bothered to examine, plus enough institutional patience to fund a quarter-century of work that produced no usable result until the very end. Once the antigen was isolated, the floodgates opened. Within five years, similar viruses had been documented across Eurasia. By 1982, the WHO had given the disease its formal name: hemorrhagic fever with renal syndrome. By 1985, the virus family had a name. By 1987, it had a genus.
For the next decade, hantavirus was an Asian and European problem. China still records tens of thousands of HFRS cases annually. South Korea produced a vaccine in 1990 — Hantavax — that works against the Asian strains. It does not work against anything else. There has never been any commercial reason to extend it.
Then in 1993, in the Four Corners region of the United States, a young Navajo couple died days apart of sudden respiratory failure, and the entire history of the disease shifted geography. CDC investigators found a new hantavirus, named it Sin Nombre — no name — after a regional dispute over what to call it. Sin Nombre did not cause kidney failure. It caused acute pulmonary failure. A new disease: hantavirus pulmonary syndrome. Mortality between 35 and 50 percent. The 1993 outbreak was driven by an El Niño year that produced a tenfold spike in deer mouse populations, which brought infected mice into closer contact with humans. The whole thing, root and branch, was the same pattern as Korea forty years earlier: rodents, climate, a quarter-century of inattention, then deaths.
Navajo oral tradition pointed to similar mass illness events in 1918, 1933, and 1934. Genetic analysis suggests Sin Nombre virus has been present in deer mice in the Americas longer than humans have lived in the Americas.
In 1995, researchers in Argentine Patagonia characterized Andes virus — a South American hantavirus carried by the long-tailed pygmy rice rat. Mortality ranged from 21 to 50 percent. In 1996, in El Bolsón, Argentina, came the discovery8 that Andes virus is the only hantavirus on Earth that can spread between humans. Every other hantavirus, in every other place, requires direct contact with rodent excreta. Andes can sometimes — rarely, requiring close prolonged contact, usually within families or healthcare settings — pass between people.
In 2012, ten people who had stayed in tent cabins in Yosemite National Park’s Curry Village contracted Sin Nombre virus. Three died. The CDC investigation found rodents had tunneled through the foam insulation of the cabins; deer mouse populations in the area had spiked that summer, with seroprevalence in trapped mice reaching 14 percent. The cabins had been there for years. The mice had been there longer. The deaths prompted the inspection.
In 2018, in a small town called Epuyén in Chubut Province, Argentina, a single index case attended a party. By the time the outbreak was over, 34 people were confirmed infected, 11 had died, and 142 had been placed in quarantine. The case fatality rate hit 32 percent.9 Argentine authorities used full-length virus sequencing to confirm what epidemiologists had suspected for two decades: that the Andes virus transmits between people during close contact, primarily during the prodromal phase, in chains that can sustain themselves through several generations before burning out. The paper documenting it was published in the New England Journal of Medicine in 2020, the same year COVID was killing millions and absorbing every dollar of pandemic preparedness funding the United States and Europe could put together. The Epuyén paper named the response framework explicitly: quarantine, contact tracing, isolation of close contacts, active clinical monitoring. This was six years before the Hondius. Sitting in a database any health ministry in the world can access for free.
By 2026, there have been roughly 3,000 documented Andes virus cases in history. Fewer than 300 documented human-to-human transmissions, ever. The CDC has logged 890 hantavirus cases in the United States between 1993 and 2023, with a 35 percent case fatality rate. Argentina has reported 101 cases since June 2025, double the prior year — a quiet doubling that nobody covered.
Most readers are encountering the word “hantavirus” for the first time this week. The reason is that hantavirus usually kills rural people in places that are not appealing to Western news desks. It took a polar expedition cruise carrying Dutch tourists, British retirees, and American passengers to register as a story.
The river in Korea did not change. The deer mice in the Four Corners did not change. The pygmy rice rats in Patagonia did not change.
VI. The funding picture
There is no Andes virus vaccine. There has never been one. Hantavax for Asian strains has existed since 1990 — thirty-six years.
There is no Andes virus antiviral. Treatment is supportive care from 1995, with better ICU technology bolted on. There is no specific therapy. There is no monoclonal antibody approved for use. There is no post-exposure prophylaxis. A patient who catches this virus and does not reach an ECMO-capable ICU within roughly 48 hours of cardiopulmonary symptoms is, statistically, on a coin flip.
The biology is well understood. Researchers at Mount Sinai, at Malbrán Institute in Buenos Aires, at USAMRIID, at the CDC’s Special Pathogens Branch all know how this virus works. The reason there is no vaccine is that there is no commercial market for one. The total addressable patient population is small, geographically concentrated in Argentina and Chile, and predominantly poor. Roughly 100 to 200 cases a year in those two countries combined. Another few hundred sporadic Sin Nombre cases in the United States. The economics do not support pharmaceutical development, and they never have.
Research has always done a poor job of prevention, because the cost of inaction is impossible to put on a balance sheet. A vaccine that prevents a million deaths over twenty years saves no specific person. There is no patient to interview. There is no story to tell. There is, politically, nothing to point at.
This is the same reason we did badly with HIV/AIDS. The Reagan administration’s funding priorities in the early 1980s were aimed elsewhere because the political cost of inaction on a disease killing gay men and IV drug users was lower than the political cost of action, and by the time the consensus shifted, the virus had a fifteen-year head start. The retroviral therapies that came later were heroic. They were also retroactive.
This is the same reason we did badly with early COVID. Coronavirus pandemic potential had been discussed in peer-reviewed literature since at least 2007 — a paper from Hong Kong called the SARS-like coronaviruses in horseshoe bats “a time bomb.” MERS appeared in 2012.10 The Ralph Baric paper showing a SARS-like bat coronavirus could infect human cells without adaptation appeared in 2015. The Global Virome Project launched and was underfunded. In 2018, the National Security Council pandemic preparedness directorate was disbanded. In October 2019, Johns Hopkins ran a tabletop simulation of a coronavirus pandemic called Event 201. Two months later, COVID emerged. The science had been published, peer-reviewed, and tabletop-rehearsed. The money to act on it had not.
The Institute for Progress (IFP) has been writing about exactly this for years. Their 2022 piece on BARDA — the Biomedical Advanced Research and Development Authority — laid it out rather well: BARDA is the federal agency tasked with developing medical countermeasures for pandemic threats. It is the agency that funded Operation Warp Speed, the $18 Billion project that got us mRNA COVID vaccines. It is, in the best honest assessment, the most effective federal agency in the COVID response. Pound for pound, it produced more output per dollar than any other arm of HHS. Almost nobody outside the field has heard of it.
BARDA’s own DRIVe program — the part of the agency designed to invest in early-stage biotech the way a venture firm would — has a budget that is, in IFP’s framing, smaller than what a single private fund deploys to biotech in a typical year. Its appropriations move in cycles tied to whatever the most recent crisis was. Congress sent $7.65 billion to fight the 2009 swine flu. It sent $32 billion in additional funds for COVID. Between those moments, the baseline budget shrinks back, talent leaves, and the agency starts the next pandemic underprepared because the political market for pandemic preparedness collapses the second the immediate threat is no longer in the news.
The IFP’s recommendation, repeated across multiple pieces, is that BARDA needs stable preparedness funding rather than emergency injections — and that the obvious, shovel-ready use of that funding is to develop prototype vaccines for each of the 26 known viral families that cause human disease before an outbreak forces a 12-month sprint. The White House Office of Science and Technology Policy estimated the cost at $24 billion. A bargain, against the trillions in social costs from a single pandemic.
That recommendation has not been funded.
BARDA has been unusually quiet about the Hondius this week. The CDC has been quieter than it should be, and that is a function of the leadership turnover that began in 2025 and is still working its way through the agency. A federal pandemic response architecture in which the agencies most equipped to lead the conversation are not leading the conversation tells you, where the system currently is.
And because the federal infrastructure has thinned, the most interesting pandemic-preparedness work in 2026 is not happening inside CDC or NIH. It is happening at organizations like ARIA — the UK’s Advanced Research and Invention Agency, modeled loosely on DARPA — where researchers like Brian Wang (see my 2025 conversation with Brian here) are running programmes on innate immunity, broad-spectrum antivirals, and pan-pathogen platforms. The thesis is that instead of developing a bespoke vaccine for every known pathogen, the human immune system itself can be tuned at the level of innate response — a kind of generic biological pre-armament that makes a population meaningfully harder to kill with any novel respiratory pathogen, before the pathogen has even been identified.
This is the kind of project that should be table stakes for a national pandemic preparedness strategy. It is, instead, happening at a small agency with a fraction of NIH’s resources. The people running these programmes are some of the best minds in biology. They are not unusual in their views. They are unusual in having institutional support to pursue them.
The reason this work is on the fringe, instead of at the core, is that traditional government science funding is built for well-defined problems with clear endpoints. It is not built for the kind of high-variance, long-horizon, “we don’t know if this will work but if it does it will save ten million lives” research. The grant cycles are too short. Peer review is too risk-averse. Success metrics are too narrow. Careers are too dependent on publishing reliable, citable work in two-year windows. The whole system selects against the kind of thinking that produced mRNA vaccines in the first place — which, remember, was fifteen years of patient, mostly-ignored, often-derided work by Katalin Karikó and Drew Weissman before anyone took it seriously.11
When the next pandemic comes — and it will come, and the timeline between major spillover events is shortening — the saving response will not come from what is inside the federal labs. It will come, if it comes, from what people at ARIA and a handful of other ‘unconventional’ scientists.12
The capital and the talent in biology in 2026 are flowing toward what can be measured in eighteen months: GLP-1 agonists, AI-driven drug discovery, longevity, peptide pharmacology, oncology. These are not bad things. Diabetes is real. Obesity is real. Cancer is real. The work is important.
The problem is that the same incentive structures that flow capital toward those things flow it away from pandemic preparedness, where the addressable market is “everyone, eventually, maybe,” the regulatory pathway is unclear because the disease does not yet exist, and the feedback loop is decades long with a feedback signal nobody can put on a quarterly earnings call.
So the work does not get done.
The soldier on the Hantan died when we did not have the knowledge or resources to save him.
Seventy-five years later, a Dutch couple died on a cruise ship in the South Atlantic from a virus in the same family. We have known what causes it for thirty years. We have known how to contain it for two decades. We have, on every available metric, no excuse.
The river in Korea did not change. The virus in the rats did not change. The thing that should have changed is us.
Will we?
Smadel, J.E., “Epidemic hemorrhagic fever,” American Journal of Public Health 43, 1327–1330 (1953).
Lee, H.W., Lee, P.W., and Johnson, K.M., “Isolation of the etiologic agent of Korean hemorrhagic fever,” Journal of Infectious Diseases 137, 298–308 (1978).
Martínez et al., “’Super-Spreaders’ and Person-to-Person Transmission of Andes Virus in Argentina,” NEJM 383:2230–2241 (2020). DOI: 10.1056/NEJMoa2009040.
R0 is pronounced R-naught. Therefore, competency R-naught.
It is worth flagging this, because the temptation to declare victory on incomplete data is the exact failure mode that produces every bad pandemic call. Eight cases, no new ones in 48 hours, the math runs forward clean. The math is wrong. We are looking at the early left tail of an incubation distribution and treating it like the right tail. The data point that matters is two weeks away.
Raymond et al., "Pathogenic hantaviruses bind plexin-semaphorin-integrin domains present at the apex of inactive, bent αvβ3 integrin conformers," PNAS 102, 1163–1168 (2005).
UK Health Security Agency, "Andes hantavirus" guidance: “The mortality rate is typically 35 to 50%.”
Wells et al., “An unusual hantavirus outbreak in southern Argentina: person-to-person transmission?” Emerging Infectious Diseases 3, 171–174 (1997).
Martínez et al., “’Super-Spreaders’ and Person-to-Person Transmission of Andes Virus in Argentina,” NEJM 383:2230–2241 (2020). DOI: 10.1056/NEJMoa2009040.
Zaki et al., “Isolation of a Novel Coronavirus from a Man with Pneumonia in Saudi Arabia,” NEJM 367, 1814–1820 (2012).
Karikó et al., “Suppression of RNA recognition by Toll-like receptors: the impact of nucleoside modification and the evolutionary origin of RNA,” Immunity 23, 165–175 (2005).
Note: ‘unconventional.’






Wow Jolie this is such an impactful piece. Your structure as well as your scientific and emotional storytelling are so powerful. I agree with you that the most important data comes later, as symptoms start to appear in patients sfter incubation time.
I also agree that we are not ready for this disease or the next pandemic. Academic and federal funding are still rewarding clear endpoint and "hot topics". I've seen a lot of virology talks with the speaker ending a slide with, "oh and the funding ran out so we stopped this line of work". Let's hope and push for a better future.
Thank you for sharing this. We need more people writing and doing God’s work.